|
The metal form (chromium as it exists in this product) is of low toxicity. The health hazards associated with exposure to chromium are dependent on its oxidation state. Other health problems that are caused by chromium(VI) are: After breathing it in chromium(VI) can cause nose irritations and nosebleeds. When it is a compound in leather products, it can cause allergic reactions, such as skin rash. People who smoke tobacco also have a higher chance of exposure to chromium.Ĭhromium(VI) is known to cause various health effects. But the uptake of too much chromium(III) can cause health effects as well, for instance skin rashes.Ĭhromium(VI) is a danger to human health, mainly for people who work in the steel and textile industry. When food in stores in steel tanks or cans chromium concentrations may rise.Ĭhromium(III) is an essential nutrient for humans and shortages may cause heart conditions, disruptions of metabolisms and diabetes. Various ways of food preparation and storage may alter the chromium contents of food. 
For most people eating food that contains chromium(III) is the main route of chromium uptake, as chromium(III) occurs naturally in many vegetables, fruits, meats, yeasts and grains. In drinking water the level of chromium is usually low as well, but contaminated well water may contain the dangerous chromium(IV) hexavalent chromium. The level of chromium in air and water is generally low. People can be exposed to chromium through breathing, eating or drinking and through skin contact with chromium or chromium compounds. Reserves are hestimated to be of the order of 1 billion tonnes with unexploited deposits in Greenland, Canada e USA. A total of 14 million tonnes of chromite ore is extracted. Chromium ores are mined today in South Africa, Zimbabwe, Finland, India, Kazakihstan and the Philippines. Chromium (IV) oxide (CrO 2) is used to manufacture magnetic tape.Ĭhromium is mined as chromite (FeCr 2O 4) ore. Chromium is used in metallurgy to impart corrosion resistance and a shiny finish as dyes and paints, its salts colour glass an emerald green and it is used to produce synthetic rubies as a catalyst in dyeing and in the tanning of leather to make molds for the firing of bricks. Chromium plating was once widely used to give steel a polished silvery mirror coating. ApplicationsĬhromium main uses are in alloys such as stainless steel, in chrome plating and in metal ceramics. Chromium is unstable in oxygen, it immediately produces a thin oxide layer that is impermeable to oxygen and protects the metal below. 
It does not tarnish in air, when heated it borns and forms the green chromic oxide. Its colour is silver-gray and it can be highly polished. Separation and Concentration Purification RequestĬhromium - Cr Chemical properties of chromium - Health effects of chromium - Environmental effects of chromiumĬhromium is a lustrous, brittle, hard metal.Plant Inspection & Process Optimalisation.You will need to refer to a periodic table for proton values. In this notation, the atomic number is not included. Symbol-mass format for the above atom would be written as Cr-52. 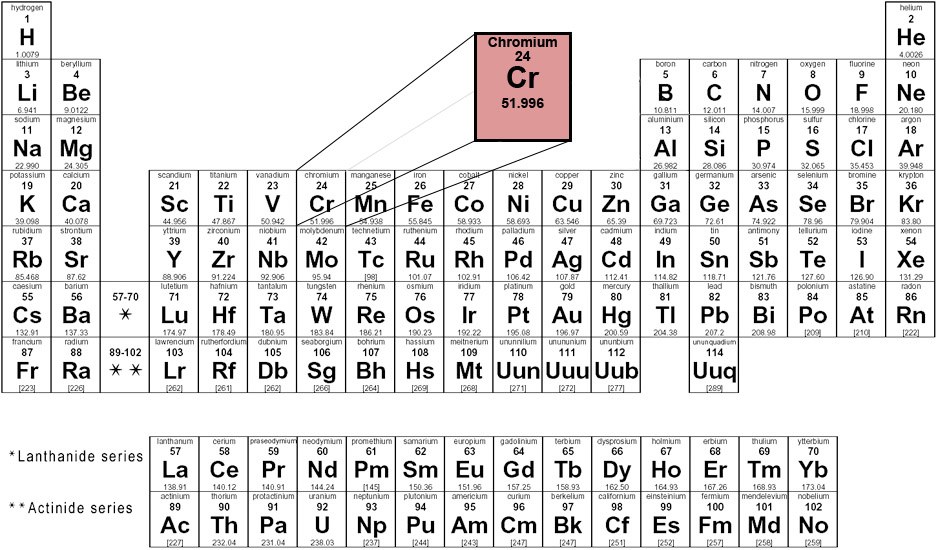
For an example of this notation, look to the chromium atom shown below:Īnother way to refer to a specific atom is to write the mass number of the atom after the name, separated by a hyphen. The "A" value is written as a superscript while the "Z" value is written as a subscript. Both the atomic number and mass are written to the left of the chemical symbol. The composition of any atom can be illustrated with a shorthand notation called A/Z format.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
